What is Starch Iodine test?
The Iodine Test for Starch is utilized to decide the presence of starch in natural materials. The test can be subjective or quantitative. The iodine test is utilized to test for the presence of starch. At the point when treated with IKI solution, iodine broke up in a watery arrangement of potassium iodide the tri-iodide-anion edifices with starch, creating a serious blue/purple coloring. The coloring can be differentiated outwardly with centralizations of iodine as low as 2×10-5 M at 20 °C. Be that as it may, the force of the colors reduces with expanding temperature and with the presence of water-miscible natural solvents, for example, ethanol. The test can’t be performed at low pH because of the hydrolysis of the starch under these conditions.
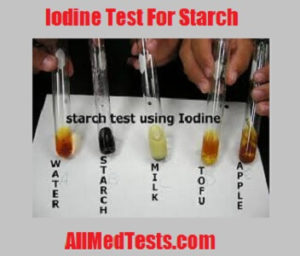
Starch Indicator:
Starch is frequently utilized as a part of science as a pointer for redox titrations where tri-iodide is available. Starch shapes an extremely dull blue-dark complex with tri-iodide which can be made by blending iodine with iodide (regularly from potassium iodide). The shade of the starch complex is deep to the point, that it can be recognized outwardly when the grouping of the iodine is as low as 0.00002 M at 20 °C. The shading affectability reduces with expanding temperature (ten times less delicate at 50 °C), and upon the expansion of natural solvents, for example, ethanol. Between iodine titrations, concentrated iodine arrangements must be reacted with some titrant, frequently thiosulfate, to uproot the greater part of the iodine before the starch is included. This is because of the insolubility of the starch-iodine complex which might keep a percentage of the iodine reacting with the titrant. Near the end-point, the starch is included, and the titration procedure is continued considering the measure of thiosulfate included before including the starch.
Principle of Iodine test for Starch:
You will be trying for the presence of this unpredictable starch or in leaves as a major aspect of a photosynthesis experiment being a Biology student. These are subjective tests. If you think about science too, you ought to focus guideline of the test, as it clarifies how starch goes about as a marker for Redox Titrations or in the ‘Iodine Clock Reaction’. These expansions of the Starch Iodine test yield subjective results.
The sole reagent required for the test is basic iodine arrangement (0.1 M potassium tri-iodide arrangement).
Procedure of Starch Iodine test:
Note: Before going down to perform the procedure for this test, you need to know that it depends on certain conditions. These conditions are whether sample used in the test is solid or a liquid. Now to make it easier for you, I am going to perform the tests in two different headings presenting for both solid and liquid. Now whether your sample is liquid or a solid, you can go for either of the test procedures.
If the sample is Solid:
- Take two fruits or vegetables because to start you will need to peel off their skin. You can take different fruits or in vegetables, Potato could be a great agent. Once you have chosen your thing, peel off their skin and get ready for the test.
- Next thing you will need to do is to remove the powdered food samples. You must do this by using a neat and clean spatula otherwise your test could be a disaster.
- Now take basic iodine solution potassium and add 2 to 3 drops of it on the food sample. Doing this on a white tile will help the purpose better because visuals are better on a white tile.
- Once being done with that, observe the results carefully and you are done.
If the sample is Liquid:
Now in the case of liquid, the test goes whole different. It still remains simple so just follow the procedure below to get positive results for the presence of starch in the case of liquid.
- Take a test tube. In the test tube, you will have to put 10 cm3 of the liquid sample of the test. Make sure that you do it carefully and nothing spills out.
- Now again in the test tube, you will have to pour down exact five drops of iodine solution and make sure that you don’t the exceed the number 5.
- Once being done with the above two steps, note down if there are any color changes appearing because this concerns the results.
Precautions before performing:
Before you perform the Iodine test for starch, here is a list of precautions that you need to take care of before and while performing.
- Make sure that amount you’re taking of samples and liquid drops are the same as mentioned in the procedure.
- Also, make sure that the test tube you are using must be cleaned and washed very neatly. There must not remain any impurity other than the natural stuff in the test tube.
- Observe colors at the end of the tests very carefully and note down results on a notepad to avoid future inconvenience.
Once taken these precautions, I am sure that your test should be 100% positive.
Results of Starch Iodine Test:
When the starch is present:
In the case of presence of starch, the color of the solution changes. The color change should be a blue-black color which appears only in the case if positive results which are that the starch is present in the solution.
When the starch is not present:
After performing the whole experiment when you are observing and no color change appears in the solution then it means that starch is not present in the solution. The result could also be negative if the experiment was not performed properly but if performed carefully and the color change doesn’t appear then it means starch is not present in the solution.
Final Words:
So this was all from a very easy test to check the vicinity of starch with the help of Iodine test. Was this article helpful in your science study? Please us your feedback and also mention if I missed something. Keep visiting our site for more amazing medical sciences tests.


Leave a Reply