Benedict’s test is utilized to test for carbohydrates and non-reducing or reducing sugar. The Benedicts test separates reducing sugars (monosaccharide’s and some disaccharides), which have free ketone or aldehyde. Benedict’s answer can be utilized to test for the presence of glucose in urine.
Test For Reducing Sugars:
A few sugars, for example, glucose are called reducing sugars since they are equipped for exchanging hydrogen (electrons) to different intensities and the procedure is called reducing. At the point when reducing sugars are blended with Benedict’s reagent and warmed, a decrease response causes Benedict’s reagent to change its coloring. The color fluctuates from green to dim red (block) or corroded cocoa, depending upon the measure of and sort of sugar.
Benedict’s quantitative reagent contains potassium thiocyanate and is utilized to decide the amount of reducing sugar which is available in the compound. This test frames a copper thiocyanate which is white and can be utilized as a part of a titration. The titration ought to be rehashed with 1% glucose arrangement rather than the specimen for adjustment.
Benedict’s reagent:
This was named after American scientist Stanley Benedict. Benedict’s reagent (frequently sold as Benedict’s Qualitative Solution or Benedict’s Solution) is a synthetic reagent.
Benedict’s reagent is a compound reagent usually used to identify the presence of reducing sugars. However, other reducing substances additionally give a positive response. This incorporates all monosaccharides and numerous disaccharides, including lactose and maltose.
For the most part, Benedict’s test will identify the presence of aldehydes, and alpha-hydroxy-ketones incorporating those that happen in certain ketones. Along these lines, despite the fact that the ketose fructose is not entirely a reducing sugar, it is an alpha-hydroxy-ketone and gives a positive test since it is changed over to the aldoses glucose and mannose by the base in the reagent. A positive test with Benedict’s reagent appears.
Benedict’s test Principle:
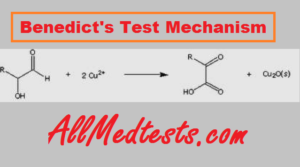
This test has actually quite a simple principle. The guideline of Benedict’s test is that when reducing sugars are warmed in the presence of an antacid, they get changed over to decreasing mixtures known as enediols. Enediols decrease the cupric particles (Cu2+) present in Benedict’s reagent to cuprous particles (Cu+) which get encouraged as insoluble red copper oxide (Cu2O).
The shade of the obtained material gives a thought regarding the amount of sugar present in the compound. The test is semi-quantitative. A greenish color demonstrates around 0.5 g% focus, the yellow result shows 1 g% fixation, orange shows 1.5 g% and red shows 2 g% or higher focus.
At the point when reducing sugars are warmed in the essential compound, they shape effective reducing mixtures known as enediol. Enderle further reacts with cupric particles which are available in Benedict’s solution for cuprous ions. Thus, we distinguish the presence of reducing compounds. Here, it should be noticed that benedict’s reagent respond with decreasing sugars as well as give a positive result with other reducing solutions.
Benedict’s test Procedure:
The procedure for this test is one of the easiest ones in the field of biological tests. With the help of little equipment, you become able to perform the test. The equipment needed for the test is a test tube, benedict’s solution, and the original solution. Once having all of this things, you can proceed with the following tests.
- Take a test tube and in it, pour down 5 ml from your portion of benedict’s solution.
- Now once you have poured down benedict’s solution, you will need to add 5 to 8 drops of the original you got in the test tube.
- Start heating the solution after it is being added in the test tube gently.
What would be the results? You are going to know this after reading the precautions.
Precaution needed to take for Benedicts test:
Before performing any scientific test, scientists have enlisted some precaution that is very necessary to be taken. If you fail to take these particular precautions, there are always chances that you test will be a failed one.
- Quantity of the benedict’s solution is very important. Before performing the test, make sure that you measure the quantity of the benedict’s solution and this that you are having it in the right amount.
- Heating the mixture should be a gentle procedure. Don’t try to be quick. Just keep it plain and gentle.
- While adding the solution, just make sure that you adding only a few drops of it. Adding the solution in more quantity could ruin the whole process.
- During the heating of the solution, make sure that it is not facing any human. Because on boiling the droplets could escape the test tube and burn the skin of any person who gets in contact.
- The last thing is very important. If you don’t get results when boiling for the first time, do it again. There are possibilities that you may not get results on the first time heating but do get after heating twice or thrice.
After all these precautions are being followed, I am sure that your results will be 100% positive.
Benedict’s test Results:
I am sorry that I had to keep you waiting for results. Now what would happen and what are going to be the results of Benedict’s test? How will you know that the test you are performing is negative or positive? Following are the results and observations.

If the sugar is reducible:
In the case when sugar is reducible, the solution will change its color after the heating process. It is not necessary that it would happen after heating just once. As mentioned in the precautions, you should try heating twice or more than it and this might bring you results.
If the sugar is non-reducible:
After heating the solution once, twice or thrice and the color remains same, then it means that the sugar in the solution is non-reducible.
Final Words:
So this was all from a very easy test to check for reducible sugar in the solution. Did our article help you in your study? Leave us your feedback in the comments section and stay tuned to our website!




It is very helpful
its very helpful indeed